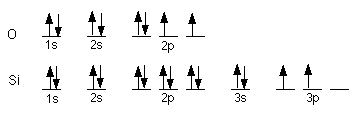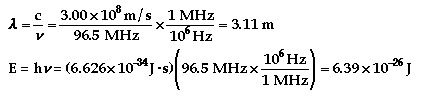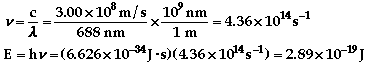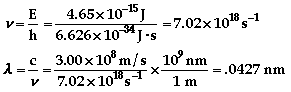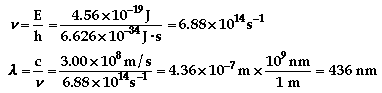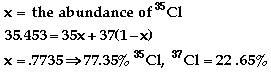Answer Key for the Practice Questions for Atomic
Theory
Go to the original worksheet.
-
a)
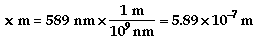
b) 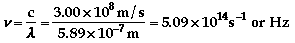
c) This is in the visible spectrum.
d)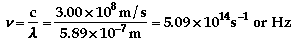
-
Quantum theory only allows for discrete energies
to be emitted, not energies at all wavelengths. The discrete energies come
from the spacing of the energy levels in which the electrons in potassium
move around. See the atomic theory
additional information for more of an explanation.
-
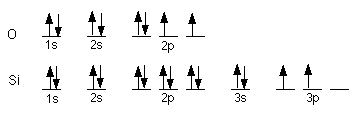
There are 2 unpaired electrons in each.
-
a) 1s22s22p1
- correct b) 1s22s13s1
excited c) 1s22s22p63s22d2
incorrect d) 1s22s42p2
incorrect e) 1s12s1excited
-
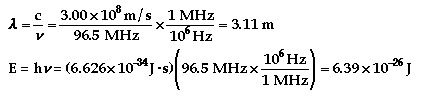
-
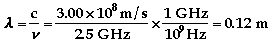
-
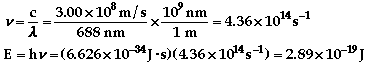
The color of light associated with 688 nm is
red.
-
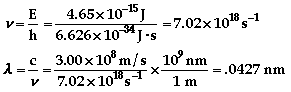
-
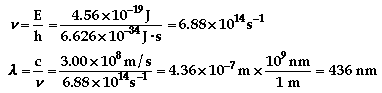
The color of light associated with 436 nm is
violet.
Answer Key for the Atom and
Atomic Theory Review Questions
-
D = m/V = 3.44 g/2.77 mL = 1.24
g/mL
-
V = m/D = 50.0 g/1.19 x 10-3
g/cm3 = 4.2 x 104 cm3.
-
For the four main ideas of Dalton's
atomic theory, see your book, p. 84.
-
Electrons were discovered by
J.J. Thomson using the cathode ray tube. See pp.86-88 for the complete
description.
-
Rutherford shot alpha particles
through thin gold foil. He expected all of the particles to go straight
through. Most did, but some deflected. This lead him to two conclusions:
first, that atoms are mostly empty space, and secondly, that there is a
positively charged, dense nucleus present. See your book for more of a
description.
-
a) Ba: p+=56, e-=
56, no= 81 b) Cu: p+= 29, e-=
29, no= 35 c) F: p+= 9, e-=
9, no= 10
-
An isotope of an element contains
the same number of protons and electrons but different number of neutrons.
-
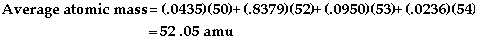
-
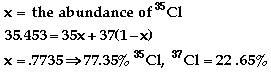
-
J.J. Thomson's model was the
"plum pudding" model - a positively charged blob with negative electrons
stuck throughout it. Rutherford stated that there was a small, dense, positively
charged nucleus with electrons outside the nucleus. Bohr also had a positive
nucleus, but stated that the electrons were in definite orbits, or energy
levels, around the nucleus. The quantum mechanical model also gives a positive
nucleus, but states that the electrons are in not in a definite orbit,
but rather in a probability region, or electron cloud.
-
An atomic orbital is the region
of space around a nucleus where an electron will most likely be (probability
region).
-
An s orbital has a spherical
shape. It holds 2 electrons. A p orbital has a dumbbell shape. It also
holds 2 electrons, however there are three p orbitals, one oriented along
the x-axis, another along the y-axis, and the third along the z-axis.
-
See your text.
-
a) S 1s22s22p63s23p4,
[Ne] 3s23p4
b) Rb 1s22s22p63s23p64s23d104p65s1,
[Kr] 5s1
c) Au 1s22s22p63s23p64s23d104p65s24d105p66s24f145d9,
though this is an exception and actually ends in 6s14f145d10,
[Xe] 6s24f145d9
-
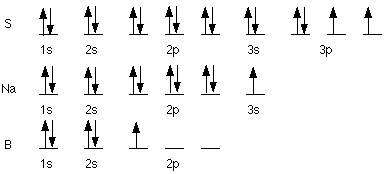
-
lowest wavelength: violet, indigo,
blue, green, yellow, orange, red: highest wavelength
-
The electron in the hydrogen
atom can be excited to various energy levels by absorbing energy. As it
relaxes back to a lower state, it releases energy. By relaxing from different
levels, different amounts of energy will be released, which produce a different
color of light. See the atomic theory
additional information for more of an explanation.
-
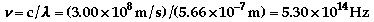
-
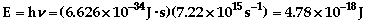
-
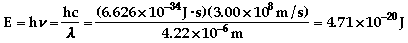
-
The ground state is the lowest energy level for an
electron to occupy. An excited state is when an electron is above the lowest
energy level available.
-
Heisenberg's uncertainty principle states that both
the position and velocity of an electron cannot be known at the same time.
-
de Broglie hypothesized that all matter can exhibit
wavelike properties. He derived an equation to describe the wavelength
of a particle
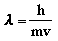 where h is Planck's constant, m is the mass of the particle, and v is its
velocity.
where h is Planck's constant, m is the mass of the particle, and v is its
velocity.
-
The photoelectric effect is where a photon (packet
of light) can eject an electron from a metal. The photon must be at least
at the threshold energy for an electron to be ejected. For a complete description,
see your book.
Notes:
E = energy (Joules)
f = frequency (hertz)
c = speed of light = 3.00
x 108 m/s
h = Planck's constant = 6.626
x 10-34 J/s
an upside down Y is lamda
= wavelength (m)
m = mass
v = velocity
Return to the Chemistry
30 outline
![]()
![]()
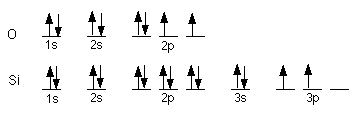
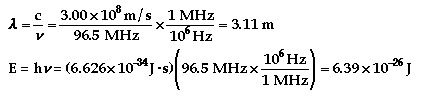
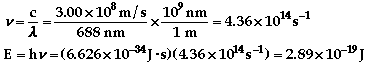
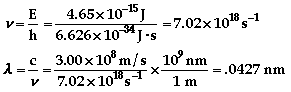
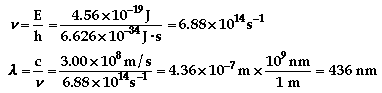
![]()
![]()