Solids usually have a definite shape and a definite volume. However, when a solid is broken into smaller pieces it is not changed chemically. For example if you crush an aspirin into a power it is still a solid just in smaller pieces.
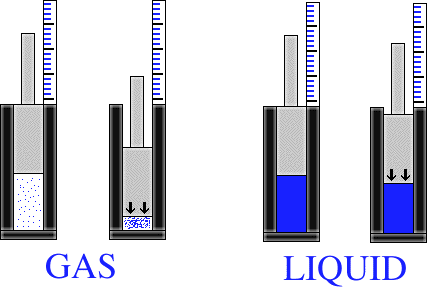
Now we have the problem of distinguishing between a liquid and a gas. What makes a liquid different from a gas is the characteristic of compressibility. A gas is easily compressible, where a liquid is not. Say for instance that you have a piston within an enclosed tube. If the tube is filled with steam, and then the piston is compressed, it is easy to compress the steam with the piston. As a result, the piston travels far into the tube. Now we put water into the enclosed tube. It is not nearly as easy to push the piston down into the tube now. Why? Well, a liquid is a lot harder to compress than a gas. This is because the molecules in the gas are farther apart than the molecules in the liquid.

These two characteristics that we have discussed, how rigid an object is, and an object's ability to be compressed, are used to determine the three basic states of matter
A solid is a form of matter which is made distinct by it rigidity. That is, a solid has a fairly fixed volume and shape, and is harder to compress than a gas or a liquid.
A liquid is a form of matter that is a fairly incompressible. This means that a liqiud basically has a fixed volume, but not a fixed shape. It takes the shape of its container.
A gas is an easily compressible fluid. This means that a given quantity of gas will fit into a container of any size and shape. A gas has neither a definite volume nor a definite shape.
Solids/Liquids/Gases | Atoms/Molecules/Ions/ | Compounds/Elements/Mixtures | Physical & Chemical Changes | Top of Page | Questions | Glossary
A molecule is a definite group of atoms that are chemically bonded together. They are tightly connected by attractive forces. A molecular formula is a chemical formula that gives the exact number of different types of atoms in a molecule. Some simple molecular substances are carbon dioxide, CO2; ammonia, NH3; and water, H2O. The atoms that are in a molecule are not just stuffed together without any order. The atoms are chemically bonded to one another in order to form a definite arrangement. A structural formula is a chemical formula which shows how the atoms are bonded to one another to form a molecule. A good example is the structural formula for water, H-O-H. Those two horizontal lines connecting the H with the O (hydrogen and oxygen) represent the chemical bonds joining the atoms.
An ion is an electrically charged particle obtained from an atom or chemically bonded group of atoms by adding or removing electrons. Now what this means is that an ion is the result of taking away, or adding, electrons to an atom or a chemically bonded group of atoms. By taking away, or adding, these electrons, the particle takes on an electrical charge. Atoms are electrically neutral as they contain an equal number of positive and negative charges. An atom that adds an extra electron to it becomes a negatively charged ion. This type of ion is called an anion. An atom which loses one or more of its electrons now has a positive charge, and is called a cation. For example, a sodium atom can lose one of its electrons and form a sodium cation. Now, instead of being Na, it would be Na+1. This means that the sodium atom has an overall positive charge of +1. Another example would be a neutral atom of Sulfur, S. If this atom of S were to gain two electrons it would become S-2. The sulfur atom would now have a total negative charge of -2.
It has 16 protons and 18 electrons.
Solids/Liquids/Gases | Atoms/Molecules/Ions | Compounds/Elements/Mixtures | Physical & Chemical Changes | Top of Page | Questions | Glossary
An element is a substance that is composed of only one kind of atom like aluminum, iron, or neon. Today, 109 elements are known and listed on the periodic table.
A compound is a substance of more than one element, chemically combined. A more scientific definition is that a compound is a type of matter composed of atoms of two or more elements chemically combined in fixed proportions. An example of a compound would be water. It is a compound that contains the elements hydrogen and oxygen fixed in the ratio 2 to 1. A compound has new properties unlike the elements which make it up. A compound has a chemical formula such as H2O.
A mixture is a material that can be separated by physical means into two or more substances. A classic example of a mixture lab would be one in which you were presented a mixture of sand, iron filings, and salt. You are told to separate these materials. How do you do that? Well, think about the various physical properties of each material. You use a magnet to separate out the iron filings. You then mix water with the sand and salt mixture. You swish the water, salt and sand around for a while and then filter it. The salt dissolved into the water, so the salt water solution passes through the filter while the sand gets left in the filter. Now we slowly heat up the salt water solution, and evaporate the water, and we are left with salt. In a mixture, the compounds are not in a definite proportion. For example, a teaspoon of salt in a liter of water is salt water, but so is a cup of salt in a liter of water.
Solids/Liquids/Gases | Atoms/Molecules/Ions | Compounds/Elements/Mixtures | Physical & Chemical Changes | Top of Page | Questions | Glossary
A chemical change is a change in which one kind of matter is changed into a different type of matter. Some examples of chemical changes: the rusting of your car, setting your shoe on fire, digesting food, and the burning of magnesium metal in oxygen to form magnesium oxide. All of these materials combine chemically with another material , and cannot be separated by any physical means.
Solids/Liquids/Gases | Atoms/Molecules/Ions | Compounds/Elements/Mixtures | Physical & Chemical Changes | Top of Page | Questions | Glossary
In chemistry and physics the idea of the atom is a key concept. To understand many of the other concepts in chemistry some knowledge of the atom is necessary. In this section the following topics will be discussed:
SubAtomic Particles | Basic Structure of an Atom | Atomic Number, Mass Number, and Isotopes | Avogadro's Number, The Mole, and Atomic Weight | Top of the page | Questions | Glossary
During the latter nineteenth century a series of experiments showed that atoms are comprised of smaller particles. An atom consists of a nucleus and one or more electrons surrounding the nucleus. The nucleus, the core of the atom, has the majority of the mass of the atom and a positive charge. An electron is a very light particle which circles the nucleus. It has a negative charge.
In an electrically neutral atom, the number of electrons equals the positive charge on the nucleus. The nucleus of the atom is composed of smaller particles called neutrons and protons. A proton has a positive charge equal in magnitude to the negative charge of an electron. This means that in an electrically neutral atom, the postive of charge the protons, combined with the negative charge of the electrons, would result in no charge because they would cancel each other out. A proton's mass, however, is a whopping 1836 times that of the electron. A neutron, however, has a mass almost identical to a proton's, but it has no electrical charge associated with it.


|
|
|
|
|
| Proton | Nucleus | 1.0073 amu | Positive |
| Neutron | Nucleus | 1.0087 amu | Neutral |
| Electrons | Electron Cloud | 0.000549 amu | Negative |
SubAtomic Particles | Basic Structure of an Atom | Atomic Number, Mass Number, and Isotopes | Avogadro's Number, The Mole, and Atomic Weight | Top of the page | Questions | Glossary
The mass number is the number of neutrons added to the number of protons. The mass number of the most common isotope can be obtained from the periodic table. If you take the decimal number on the periodic table and round it to the nearest whole number, you have the mass number. For example the atomic weight of Iron(Fe) is 55.847. When rounded it gives a mass number of 56.
The atomic number of Fe is 26. so most Fe atoms have 30 (56-26) neutrons. In addition, all neutral Fe atoms have 26 protons and 26 electrons. Atoms of the same element with a different number of neutrons are called isotopes. The most common isotope of an element is the one that is on the periodic table.
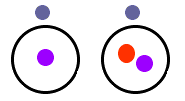

The above graphic shows two isotopes of Hydrogen. The picture on the left is the most common isotope of hydrogen with one electron and one proton. The picture on the right is another isotope of hydrogen with one proton, one electron, and a neutron. The most common isotope of uranium is uranium-238 which has 92 protons, 92 electrons, and 146 neutrons. Another isotope is uranium-235 with 92 protons, 92 electrons, and 143 neutrons.
SubAtomic Particles | Basic Structure of an Atom | Atomic Number, Mass Number, and Isotopes | Avogadro's Number, The Mole, and Atomic Weight | Top of the page | Questions | Glossary
Probably the only thing you will ever have a mole of is atoms or molecules. One mole of magnesium atoms (6.02 x 1023 magnesium atoms) weigh 24.3 grams. 6.02 x 1023 carbon atoms weigh a total of 12.0 grams. 6.02 x 1023 molecules of CO2 gas only weigh a total of 44.0 grams. The decimal number on the periodic table is the atomic mass, the mass of one atom measured in atomic mass units(amu). Amu's are defined to be 1/12 the weight of the most common isotope of Carbon. This number in grams is the mass of 1 mole of that element. For example, 6.02 x 1023 iron atoms weigh only 55.847 grams.(This is equivalent to saying one mole of iron atoms weighs 55.847 grams.) One mole of sulfur weighs 32.066 grams. (This is the same as saying 6.02 x 1023 Sulfur atoms weigh 32.066 grams)
When not measured in grams, the decimal number on the periodic table is called the atomic mass and is in atomic mass units(amu). As mentioned earlier, one proton weighs 1.0073 amu and 1 neutron weighs 1.0087 amu. So the atomic mass is the mass in amus of one atom of an element, but you rarely use the mass of one atom. Even if you have a tiny speck of a metal or a microgram of an element, you have billions and billions of atoms. Thus, the mass in grams of one mole of an element (the gram atomic weight) is more useful.
SubAtomic Particles | Basic Structure of an Atom | Atomic Number, Mass Number, and Isotopes | Avogadro's Number, The Mole, and Atomic Weight | Top of the page | Questions | Glossary

The final quantum number is the spin quantum number, it describes the spin orientation of an electron.
The electron configuration of an atom is the particular distribution of electrons among available shells. It is described by a notation that lists the subshell symbols, one after another. Each symbol has a subscript on the right giving the number of electrons in that subshell. For example, a configuration of the lithium atom (atomic number 3) with two electrons in the 1s subshell and one electron in the 2s subshell is written 1s22s1.
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
The notation for electron configuration gives the number of electrons in each subshell. The number of electrons in an atom of an element is given by the atomic number of that element.
On the left we have a diagram to show how the orbitals of a subshell
are occupied by electrons. On the right there is a diagram for the filling
order of electrons in a subshell.

Here are some examples that show how to use the filling order diagram to complete the electron configuration for a certain substance.
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
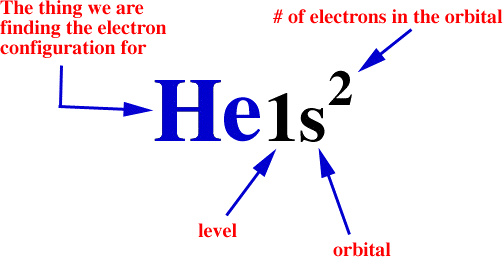
Often times you will be asked to find the electron configuration for something that looks like this:
53I
The 53 denotes the number of electrons in an atom of iodine. You would now proceed to do the electron configuration by looking at the filling order chart.
1s22s22p63s23p64s2 3d104p65s24d105p5
Electronic Structure of Atoms | Periodicity | Top of Page | Questions | Glossary
Electronic Structure of Atoms | Periodicity | Top of Page | Questions | Glossary